by Jason Wasserman MD PhD FRCPC
April 7, 2026
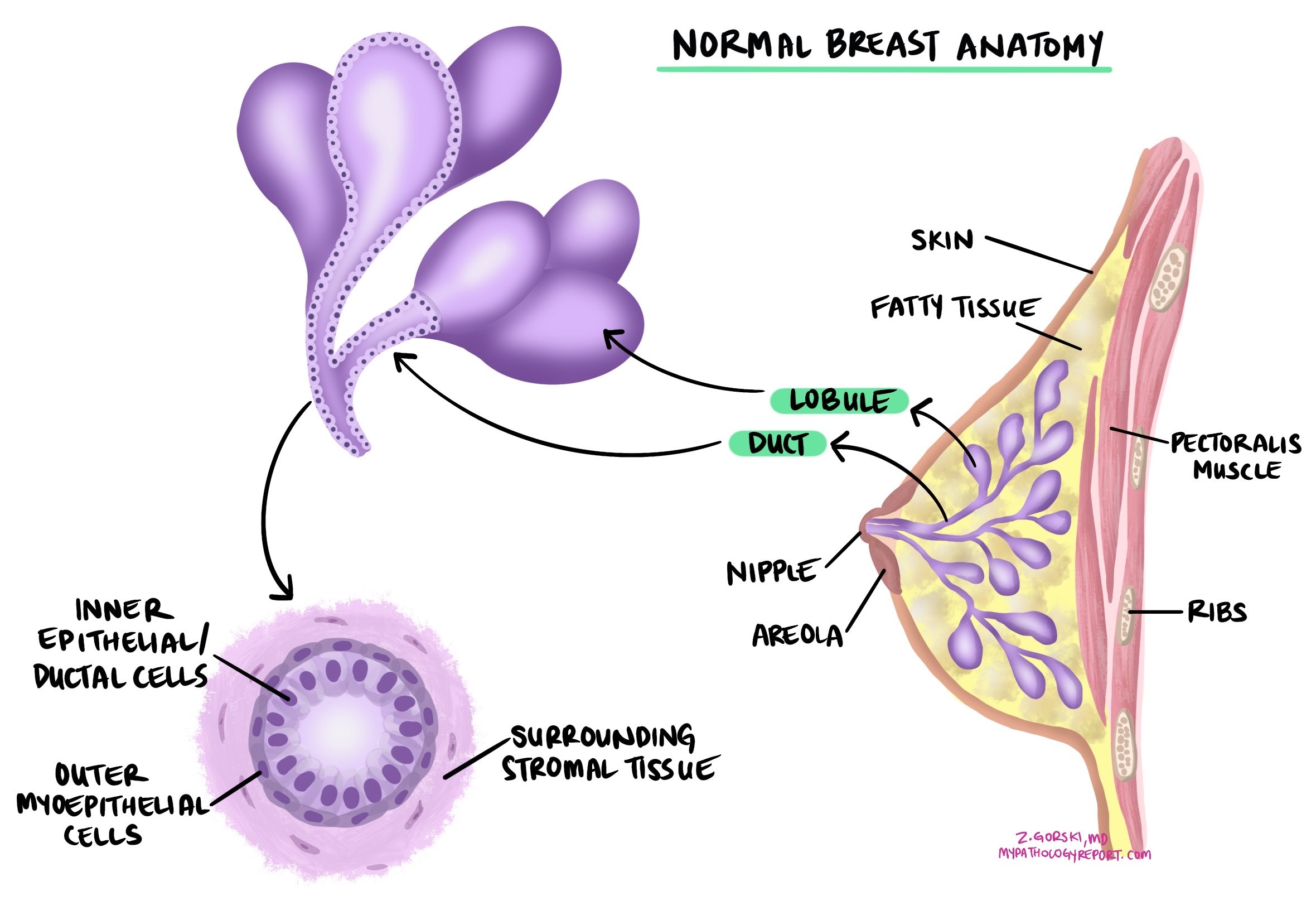
Ductal carcinoma in situ (DCIS) is a noninvasive type of breast cancer that starts in the lining of the breast ducts — the tiny tubes that carry milk from the glands to the nipple. Noninvasive means that all the abnormal cells are confined inside the ducts and have not broken through the duct wall into nearby breast tissue. Because the cells are contained, DCIS cannot spread to lymph nodes or other organs unless it first becomes invasive. Treatment aims to remove it completely and prevent it from becoming invasive.
This article will help you understand the findings in your DCIS pathology report — what each term means and why it matters for your care. If you had a breast biopsy, you may also find our guide to understanding your breast biopsy report helpful.
What are the symptoms?
Most people with DCIS have no symptoms. It is usually found on a screening mammogram, often appearing as tiny calcium deposits called microcalcifications. Less commonly, someone may notice a lump, nipple discharge (which can be bloody), or subtle changes in breast shape or texture.
What causes DCIS?
DCIS develops when breast duct cells acquire DNA changes that cause them to grow and divide too quickly. Risk is influenced by many of the same factors as invasive ductal carcinoma, including inherited gene variants (such as BRCA1/BRCA2), family history of breast cancer, long lifetime exposure to estrogen (from factors such as later menopause or hormone replacement therapy), higher breast density, obesity, alcohol use, and prior chest radiation. Most cases arise without an identifiable inherited cause.
How is the diagnosis made?
DCIS is usually diagnosed on a core needle biopsy performed because of an abnormal finding on a mammogram, most often microcalcifications. A pathologist confirms DCIS by examining the tissue under the microscope and identifying abnormal cells confined entirely within the breast ducts and glands. The key criterion is that the duct wall — supported by a layer of cells called myoepithelial cells — remains intact around the abnormal cells. If any cancer cells are found outside the duct wall and growing into the surrounding breast tissue, the diagnosis changes to invasive ductal carcinoma.

After the biopsy confirms DCIS, surgery (lumpectomy or mastectomy) is typically performed to remove the remaining disease. The surgical specimen is then examined in full, allowing the pathologist to assess the complete size, grade, and margin status.
What is the risk of DCIS progressing to invasive cancer?
DCIS is confined to the ducts, but some cases can progress to invasive ductal carcinoma if not treated. After treatment, cancer can recur in the same breast as DCIS again or, less commonly, as invasive cancer. The risk of recurrence and progression depends on several features reported in the pathology report — particularly nuclear grade, the presence of comedonecrosis, margin status, and the extent (size) of the DCIS — as well as treatment choices such as surgery type, radiation therapy, and endocrine therapy. The risk of cancer developing in the opposite breast is only slightly increased compared with the treated breast.
What stage is DCIS?
Because all the cancer cells are confined to the ducts and glands and have not spread into the surrounding breast tissue, DCIS is always assigned the stage pTis (carcinoma in situ). Stage does not vary by size or grade of DCIS — all DCIS is pTis. However, size and grade are still very important because they affect recurrence risk and treatment decisions, even though they don’t change the stage.
Nuclear grade
Nuclear grade describes how abnormal the cancer cell nuclei look under the microscope and how actively the cells are dividing. It is one of the most important predictors of the risk of recurrence and progression in DCIS. The nucleus is the control center of the cell — it contains the DNA and directs cell behavior.
- Low nuclear grade (grade 1) — Cells have small, uniform nuclei that look relatively close to normal. Mitotic figures (cells visibly dividing under the microscope) are rare. Low-grade DCIS is less likely to recur or progress to invasive cancer if completely treated.
- Intermediate nuclear grade (grade 2) — Cells show moderate enlargement and variation in nuclear size and shape. Mitotic activity is present but limited. Risk of recurrence sits between low and high grade.
- High nuclear grade (grade 3) — Cells have large, irregular, pleomorphic nuclei and frequent mitoses. High-grade DCIS is more often associated with comedonecrosis (dead cells in the duct center) and carries a higher risk of recurrence and progression to invasive cancer without appropriate treatment.
Your report may also explicitly include the mitotic rate. A higher mitotic rate generally correlates with higher grade and greater recurrence risk.
Histologic subtypes
DCIS is also described by the structural pattern formed by the abnormal cells within the ducts. These patterns appear in the pathology report and help with radiology-pathology correlation and risk estimation. Treatment decisions are guided by the overall picture — grade, size, necrosis, and margin status — not the subtype alone.
- Solid pattern — Cancer cells fill the duct from wall to wall with no spaces between them. This pattern is common in high-grade DCIS but can occur at any grade. On imaging, it may appear as linear or segmental calcifications.
- Cribriform pattern — Cells form evenly spaced round holes within the duct, giving it a “Swiss cheese” appearance. This pattern is often low- or intermediate-grade and may produce granular or amorphous calcifications on mammography.
- Micropapillary pattern — Tiny finger-like tufts of cells project into the duct without a central blood-vessel core. Micropapillary DCIS can be extensive and is sometimes linked with a higher local recurrence risk, particularly when high-grade or large in extent.
- Papillary pattern — Branching fronds of cells protrude into the duct, each supported by a delicate blood-vessel core. Pathologists distinguish papillary DCIS from benign intraductal papilloma based on cellular features and special stains. Papillary DCIS may form a palpable mass but is still in situ as long as the duct wall is intact.
Comedonecrosis
Comedonecrosis is a term pathologists use to describe dead cancer cells visible in the center of a duct affected by DCIS. Under the microscope, this appears as a plug of cellular debris — the remnants of tumor cells that have died, sometimes with calcium deposits. Your report will state whether comedonecrosis is present or absent.
Comedonecrosis is important because it most often occurs in high-grade DCIS and is associated with a higher risk of local recurrence after treatment and a higher chance of progression to invasive ductal carcinoma if left untreated. When comedonecrosis is reported, it usually indicates a more aggressive form of DCIS that warrants close attention in treatment planning. Comedonecrosis is a form of necrosis specific to ductal in situ disease.
Size and extent
The size (extent) of DCIS describes how much of the breast is involved by the abnormal cells. Although size does not change the official stage — all DCIS is pTis — it is critical for treatment planning. The extent of DCIS helps estimate the risk of recurrence, the likelihood that invasive cancer is present nearby, and whether breast-conserving surgery (lumpectomy) is achievable with clear margins.
Why is size difficult to measure in DCIS?
Unlike invasive cancers, which typically form a solid mass, DCIS grows within the branching network of milk ducts. The abnormal cells may spread across a large area in a complex three-dimensional pattern that is difficult to capture on a single microscope slide. Several additional factors make measurement challenging:
- DCIS often grows in an irregular, branching pattern that may skip areas between involved ducts.
- Breast tissue is soft and compressib so thatso the apparent size may differ between surgery, specimen imaging, and laboratory processing.
- DCIS may not be removed in one continuous piece, especially when it involves a large area.
- Imaging (such as mammography) shows calcifications associated with DCIS, but sometimes underestimates or overestimates the true extent.
How do pathologists estimate size?
Pathologists use several methods depending on how the tissue is submitted:
- Single-slide measurement — If DCIS is confined to a single tissue block, its size can be measured directly. This works best for small, localized lesions.
- Serial sequential sampling — The entire specimen is mapped, serially sliced, and the extent is reconstructed across all pieces. This gives the most accurate picture of widespread DCIS.
- Block counting — When DCIS appears in multiple tissue blocks, the number of involved blocks is multiplied by the block thickness to estimate the total size.
- Opposite margin distance — When DCIS is present at or near two opposite edges of the specimen, the distance between those edges provides a minimum size estimate.
Pathologists report the largest estimate obtained using these methods.
Why does size matter for treatment?
- Up to 20 mm — Breast-conserving surgery with clear margins is generally achievable.
- 20–40 mm — Wide clear margins may be harder to obtain; re-excision is more likely to be needed.
- More than 40 mm — Breast-conserving surgery may not be possible, and mastectomy may be recommended. To a greater extent, it also increases the likelihood that an invasive cancer focus is present in the specimen.
Surgical margins
A margin is the edge of tissue removed during surgery. The pathologist examines the cut surfaces to determine whether DCIS cells are present at the edge, which indicates whether the disease was completely excised.
- Negative (clear) margin — No DCIS at the cut edge. For DCIS treated with lumpectomy, a margin of at least 2 mm is generally considered adequate. A wider negative margin is associated with a lower risk of local recurrence.
- Close margin — DCIS is within 2 mm of the cut edge but does not reach it. This may warrant consideration of re-excision or radiation therapy, depending on the grade and clinical context.
- Positive margin — DCIS cells are present at the cut edge, suggesting the disease extends beyond the resection. Additional surgery (re-excision or mastectomy) is typically recommended.
Note that the 2 mm threshold applies specifically to DCIS treated by lumpectomy without planned radiation. When radiation therapy is part of the plan, narrower margins may be acceptable. Your surgeon and radiation oncologist will interpret the margin results in context.
Biomarker and molecular testing
Estrogen receptor (ER) and progesterone receptor (PR)
Hormone receptors are proteins found in some breast cancer cells that allow them to respond to estrogen and progesterone. The two main types are estrogen receptor (ER) and progesterone receptor (PR). Testing for ER and PR in DCIS helps guide decisions about whether endocrine (hormone-blocking) therapy is appropriate after surgery. Results are reported by immunohistochemistry (IHC) and may include:
- Percentage of positive cells — For example, “80% ER-positive” means 80% of cancer cells express the estrogen receptor.
- Staining intensity — Reported as weak, moderate, or strong.
- Overall score (Allred or H-score) — Combines percentage and intensity into a single number; higher scores indicate stronger receptor expression.
DCIS is considered hormone receptor-positive if ER or PR is present in at least 1% of cells. Hormone receptor-positive DCIS may benefit from adjuvant endocrine therapy — typically tamoxifen or an aromatase inhibitor — to reduce the risk of recurrence in the treated breast and of new cancer developing in the opposite breast. DCIS with ER positivity between 1% and 10% is described as ER-low positive; these cases generally still benefit more from endocrine therapy than ER-negative DCIS.
HER2
HER2 testing is not routinely performed on all DCIS, but it may be included in the pathology report, particularly for high-grade cases or as part of a research protocol. When tested, results are scored using the same IHC scoring system (0, 1+, 2+, 3+) as for invasive breast cancer, with equivocal 2+ results confirmed by fluorescence in situ hybridization (FISH). High-grade DCIS is more likely to be HER2-positive than low-grade DCIS. HER2 status in DCIS does not currently drive targeted treatment decisions the way it does in invasive cancer, but it may inform discussions about risk and the likelihood of invasive recurrence.
Genomic testing
A genomic test called the DCIS Score (Oncotype DX DCIS) is available for hormone receptor-positive DCIS treated by lumpectomy. It analyzes the activity of 12 genes in the tumor to estimate the 10-year risk of local recurrence — either as DCIS again or as invasive cancer — after lumpectomy alone (without radiation). The score helps guide decisions about whether radiation therapy is likely to provide meaningful benefit.
The DCIS Score is not appropriate for all patients — it is validated only in hormone receptor-positive DCIS treated by lumpectomy — and its use depends on individual clinical factors. Your oncologist will discuss whether it applies to your situation. For more information about breast cancer biomarkers, visit our Biomarkers and Molecular Testing section.
What is the prognosis for DCIS?
The prognosis for DCIS is excellent. Because DCIS is a noninvasive cancer that has not spread beyond the ducts, it is associated with very high long-term survival rates when treated appropriately. DCIS itself is not life-threatening — the goal of treatment is to prevent it from progressing to invasive breast cancer, which can spread to other parts of the body.
The risk of recurrence or progression after treatment varies depending on the specific features reported in your pathology report:
- Nuclear grade — High-grade DCIS has a higher recurrence risk than low-grade DCIS. High-grade disease with comedonecrosis carries the greatest risk of becoming invasive if undertreated.
- Margin status — Negative margins with at least 2 mm clearance are associated with the lowest rates of local recurrence. Positive or close margins substantially increase the risk of recurrence.
- Size and extent — Larger DCIS is associated with higher recurrence risk, partly because it may be more difficult to remove completely.
- Treatment — Lumpectomy alone, lumpectomy plus radiation, and mastectomy are all options depending on the above features. Adding radiation after lumpectomy roughly halves the rate of local recurrence. Adding endocrine therapy for hormone receptor-positive DCIS further reduces risk in the treated and opposite breast.
Most people treated for DCIS do not develop invasive breast cancer. However, long-term follow-up is recommended because new DCIS or invasive cancer can develop years after initial treatment, in either the treated or the opposite breast.
Questions to ask your doctor
Your pathology report contains important information that will guide your care. The following questions may help you prepare for your next appointment.
- Is my DCIS low, intermediate, or high-grade nuclear?
- Was comedonecrosis present, and how does that affect my risk?
- What histologic pattern was reported, and how extensive is the DCIS?
- What is the size or extent of the DCIS, and does it affect whether lumpectomy is possible?
- Were the surgical margins clear? If not, what are my options — re-excision or mastectomy?
- What is the distance from the DCIS to the nearest margin, and is it adequate?
- Is my DCIS ER-positive, and would endocrine therapy (tamoxifen or an aromatase inhibitor) help reduce my recurrence risk?
- Was HER2 testing performed, and what were the results?
- Would the DCIS Score (Oncotype DX) test help decide whether I need radiation therapy?
- What is my estimated risk of the DCIS coming back or progressing to invasive cancer, based on my pathology report?
- What additional treatment is recommended after surgery — radiation therapy, endocrine therapy, or both?
- What follow-up imaging and appointments will I need, and how often?


