by Jason Wasserman MD PhD FRCPC and Zuzanna Gorski MD FRCPC
April 6, 2026
Invasive ductal carcinoma is the most common type of breast cancer. It starts in the epithelial cells lining the milk ducts of the breast and grows into the surrounding breast tissue. Left untreated, it can spread to other parts of the body, including the lymph nodes, bones, and lungs. The term “invasive breast carcinoma” is another name for this diagnosis.
This article will help you understand the findings in your pathology report — what the terms mean, what the numbers indicate, and why each piece of information matters for your care. If you had a breast biopsy or surgery, you may also find our guide to understanding your breast biopsy report helpful.
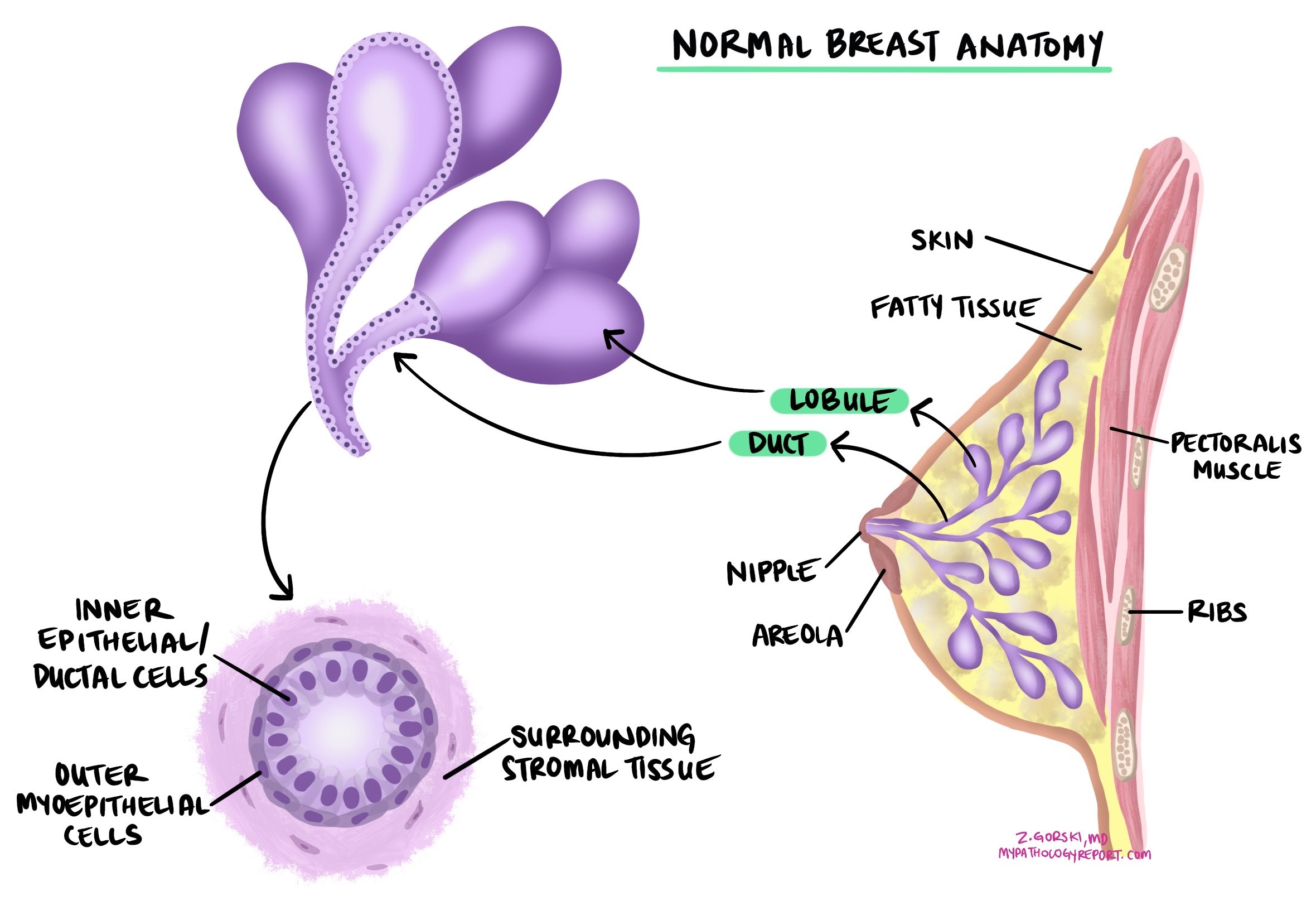
What causes invasive ductal carcinoma?
The exact cause is not known, but several factors increase the risk. Inherited gene mutations — particularly in BRCA1 or BRCA2 — significantly raise the risk of breast cancer, especially in people with a family history. Hormonal factors also play a role: starting menstruation early, going through menopause late, having no children or a first child after age 30, and using hormone replacement therapy are all associated with increased risk.
Risk also rises with age, especially after 50. A personal history of breast cancer or precancerous conditions such as atypical ductal hyperplasia, or a prior diagnosis of ductal carcinoma in situ (DCIS) — a non-invasive form of breast cancer confined to the ducts — increases the chance of developing invasive cancer. Prior chest radiation, especially in childhood or young adulthood, is another established risk factor. Lifestyle factors, including alcohol use, obesity, and physical inactivity, also contribute.
What are the symptoms?
The most common symptom is a new lump or mass in the breast, which is often hard and irregular in shape but can also be soft or round. Other symptoms include changes in the size or shape of the breast, dimpling or redness of the skin, nipple inversion or discharge (especially bloody discharge), persistent pain in one area of the breast, and swelling of the breast without a lump. Enlarged lymph nodes under the arm or near the collarbone may also be signs of invasive ductal carcinoma.
How is the diagnosis made?
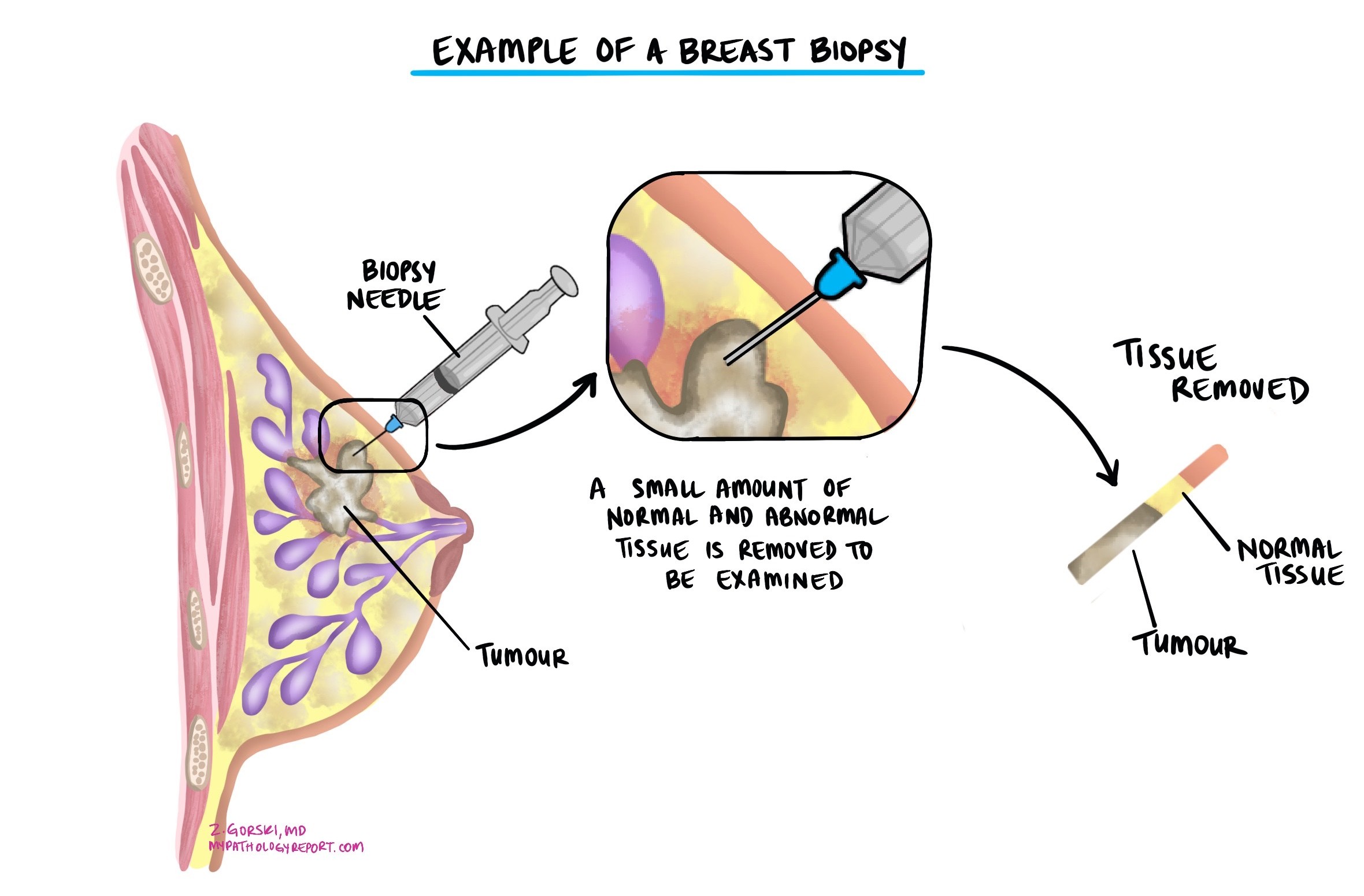
The diagnosis is usually made after a small sample of the tumor is removed in a procedure called a biopsy and examined under a microscope by a pathologist. Additional surgery to remove the entire tumor is typically recommended after the biopsy confirms cancer.
Nottingham histologic grade
The Nottingham histologic grade (also called the modified Scarff-Bloom-Richardson grade) is one of the most important findings in a breast cancer pathology report. It describes how abnormal the cancer cells look and how quickly they are growing — information used to predict how aggressive the tumor is likely to be and to guide treatment decisions.
The grade is calculated by scoring three microscopic features, each on a scale of 1 to 3:
- Tubule formation — This measures how much of the tumor forms round, gland-like structures called tubules, which resemble normal breast tissue. A score of 1 means most cells form tubules; a score of 3 means very few tubules are present.
- Nuclear pleomorphism — This describes how variable and abnormal the nuclei (the center of each cell) look compared to normal breast cells. A score of 1 means the nuclei are relatively uniform; a score of 3 means they are markedly enlarged and irregular.
- Mitotic count — This counts how many cells are actively dividing (mitotic figures) in a defined area of the tumor. A higher count means the tumor is growing more rapidly.
The three scores are added together (total range: 3 to 9) to determine the overall grade:
- Grade 1 (low grade) — Total score 3–5. Cancer cells closely resemble normal cells and tend to grow slowly. Associated with a more favorable outlook.
- Grade 2 (intermediate grade) — Total score 6–7. Cancer cells show moderate abnormality and grow at an intermediate rate.
- Grade 3 (high grade) — Total score 8–9. Cancer cells look distinctly abnormal and tend to grow rapidly. Associated with a higher risk of spread and may require more aggressive treatment.
Ductal carcinoma in situ (DCIS) in the same specimen
Your pathology report may mention ductal carcinoma in situ (DCIS) alongside the invasive cancer. DCIS refers to abnormal cells confined to the milk ducts that have not yet broken into the surrounding breast tissue. Finding DCIS near invasive ductal carcinoma is common and supports the understanding that the invasive cancer developed from a pre-existing in situ lesion. The extent of DCIS (how large an area it involves) and its grade may be reported separately, as these factors can influence surgical decisions.
Micropapillary features
Your report may note that the tumor has micropapillary features, meaning small clusters of tumor cells appear to float in open spaces under the microscope. This growth pattern is significant because tumors with micropapillary features are more likely to invade nearby lymphatic vessels and spread to lymph nodes. When more than 90% of the tumor shows this pattern, it is classified as a separate entity called invasive micropapillary carcinoma, which may have specific treatment implications. Studies show these tumors have a higher chance of axillary lymph node involvement. However, this does not necessarily worsen long-term survival when the cancer is compared stage-for-stage with other invasive ductal carcinomas.
Mucinous features
When the tumor cells are surrounded by large amounts of mucin (a gel-like substance), your report may describe mucinous features. If more than 90% of the tumor is mucinous, it is classified as invasive mucinous carcinoma — a distinct subtype with a generally more favorable prognosis that tends to grow slowly and is less likely to spread to lymph nodes. When the tumor has a mix of mucinous and non-mucinous areas, the behavior depends on the proportions and other tumor characteristics.
Tumor size
Tumor size is one of the most important factors in breast cancer, used both to determine the pathologic tumor stage (pT) and because larger tumors are more likely to metastasize to lymph nodes and other organs. The final tumor size can only be accurately measured after the entire tumor has been removed surgically — it is not included in a biopsy report, only in the surgical specimen report.
Tumor extension
Invasive ductal carcinoma begins in the breast, but in some cases the tumor spreads into the overlying skin or the muscles of the chest wall. This is called tumor extension. Its presence is associated with a higher risk of local recurrence and distant spread, and it raises the pathologic tumor stage (pT4).
Lymphovascular invasion
Lymphovascular invasion (LVI) means cancer cells have entered small blood vessels or lymphatic channels near the tumor. These vessels can act as highways for cancer cells to travel to lymph nodes or other organs. Your report will describe this as “present” (or “positive”) or “absent” (or “negative”). When lymphovascular invasion is present, the risk of spread and recurrence is higher, and your doctor may recommend additional treatment such as chemotherapy or radiation therapy.
Surgical margins
A margin is the edge of tissue removed during surgery. The pathologist examines the margins to determine whether the entire tumor was removed.
- Negative margin — No cancer cells are seen at the cut edge. This is the goal of surgery and is associated with a lower risk of local recurrence.
- Positive margin — Cancer cells are present at the cut edge, meaning some cancer may remain. Additional surgery or radiation is usually recommended.
Even when all margins are negative, the report may include a measurement of how close the nearest tumor cells came to the edge — a wider negative margin generally lowers the risk of recurrence. Margins are assessed only after surgery that removes the entire tumor, not after a biopsy.
Lymph nodes
Lymph nodes are small immune organs that filter fluid and can trap cancer cells. When breast cancer spreads, it often travels first to the axillary lymph nodes (under the arm). During surgery, some of these nodes are removed and examined under the microscope. Your pathology report will include the number of lymph nodes examined, the number that contain cancer, and the size of any cancer deposits.
There are three levels of lymph node involvement:
- Isolated tumor cells (ITCs) — Tiny clusters no larger than 0.2 mm. These are not counted as positive for staging purposes and generally have little impact on treatment decisions.
- Micrometastasis — Cancer clusters between 0.2 mm and 2 mm. Reported as pN1mi. May slightly increase the risk of recurrence and influence treatment decisions.
- Macrometastasis — Cancer clusters larger than 2 mm. Indicates a higher risk of spread and typically leads to more intensive treatment recommendations, such as chemotherapy or radiation to the lymph node region.
Your report may also mention extranodal extension, meaning cancer has broken through the outer wall of a lymph node into the surrounding tissue — a finding associated with higher recurrence risk. The terms sentinel lymph node (the first lymph node in the drainage chain from the breast) and non-sentinel axillary lymph node (those further along the chain) may also appear.
Biomarker and molecular testing
Biomarker testing is an essential part of every breast cancer workup. The results directly determine which treatments are most likely to be effective and help estimate the risk of recurrence.
Estrogen receptor (ER) and progesterone receptor (PR)
Estrogen receptor (ER) and progesterone receptor (PR) are proteins found in some breast cancer cells that allow them to respond to the hormones estrogen and progesterone. Tumors that express these receptors use the hormones to fuel their growth. Testing for ER and PR is performed by immunohistochemistry on a biopsy or surgical specimen.
Your report will include:
- Percentage of positive cells — For example, “80% ER-positive” means 80% of cancer cells express the estrogen receptor.
- Staining intensity — Described as weak, moderate, or strong, reflecting the amount of receptor protein present.
- Overall score — Some reports use the Allred score or H-score, whichcombinese percentage and intensity into a single number.
A cancer is considered hormone receptor-positive if ER or PR is present in at least 1% of cells. These cancers tend to grow more slowly and typically respond well to hormone-blocking therapies such as tamoxifen or aromatase inhibitors (anastrozole, letrozole, exemestane), which reduce the chance of recurrence. Tumors with ER positivity between 1% and 10% are considered ER low positive — they generally still benefit from hormone therapy more than ER-negative cancers.
When ER, PR, and HER2 are all negative, the tumor is called triple-negative breast cancer. Triple-negative cancers do not respond to hormone therapy or HER2-targeted drugs; chemotherapy is the mainstay of systemic treatment, though immunotherapy (pembrolizumab) is now approved for certain triple-negative cases.
HER2
HER2 (human epidermal growth factor receptor 2) is a protein that helps control cell growth. In some breast cancers, the HER2 gene is amplified, meaning extra copies are present and the cancer cells produce too much HER2 protein. These tumors — called HER2-positive — often grow more quickly but respond very well to HER2-targeted therapies such as trastuzumab (Herceptin), pertuzumab, and trastuzumab-deruxtecan.
HER2 is tested in two steps:
Step 1: Immunohistochemistry (IHC) measures the amount of HER2 protein on the surface of tumor cells and is reported as a score:
- 0 (negative) — No visible membrane staining. The tumor is HER2-negative. Targeted HER2 therapies are not effective.
- 0+ (ultralow) — Faint or barely visible incomplete membrane staining in 10% or fewer tumor cells. Still considered HER2-negative by standard criteria, but emerging research suggests these tumors may respond to newer antibody-drug conjugates in the metastatic setting.
- 1+ (low) — Faint, incomplete membrane staining in more than 10% of cancer cells. Classified as HER2-low. Considered HER2-negative by traditional criteria, but HER2-low cancers may be eligible for trastuzumab-deruxtecan in the metastatic setting.
- 2+ (equivocal) — Weak to moderate complete membrane staining in more than 10% of tumor cells. The result is borderline; further testing with in situ hybridization (ISH) is required. If ISH is negative, the tumor is reclassified as HER2-low.
- 3+ (positive) — Strong, complete membrane staining in more than 10% of cancer cells. The tumor is HER2-positive. HER2-targeted therapies are recommended.
Step 2: In situ hybridization (ISH) is performed when the IHC result is 2+ (equivocal). This test counts the number of HER2 gene copies inside tumor cells using fluorescent or silver stains and compares them to a reference gene (CEP17). Results are reported as a ratio and signal count:
- Positive (amplified) — Extra copies of the HER2 gene confirm a HER2-positive tumor.
- Negative (not amplified) — Normal gene copy number confirms the tumor is HER2-negative (reclassified as HER2-low if IHC was 1+ or 2+).
Your report may describe the ISH result using one of five groups (Groups 1–5) defined by international guidelines. Groups 2, 3, and 4 are equivocal and require interpretation in conjunction with the IHC score. Your pathologist integrates both results to give the final HER2 classification.
Genomic testing (gene expression profiling)
In addition to protein-based biomarker tests, some patients with hormone receptor-positive, HER2-negative early breast cancer receive genomic tests that analyze the activity of specific genes in the tumor to estimate the risk of recurrence and predict the benefit of chemotherapy. These results may appear in your pathology report or be reported separately.
- 21-gene recurrence score (Oncotype DX) — Analyzes 21 genes and generates a score from 0 to 100. A low score suggests a low risk of recurrence and indicates that chemotherapy is unlikely to add benefit beyond hormone therapy. A high score suggests a higher risk and that chemotherapy is likely to provide additional benefit.
- 70-gene signature (MammaPrint) — Classifies tumors as “low risk” or “high risk” for distant recurrence, also helping to determine whether chemotherapy is needed.
Your oncologist will explain whether genomic testing applies to your situation and how the result affects your treatment plan. For more information about breast cancer biomarkers, visit our Biomarkers and Molecular Testing section.
Treatment effect and residual cancer burden
If you received chemotherapy, targeted therapy, or hormone therapy before surgery (called neoadjuvant therapy), your pathology report will describe how much tumor remains in the breast and lymph nodes — known as the treatment effect.
The most commonly used measure is the Residual Cancer Burden (RCB) index, which combines the size of the tumor bed, the percentage of remaining cancer cells, and the extent of lymph node involvement into a single score:
- RCB-0 (pathologic complete response) — No residual invasive cancer in the breast or lymph nodes. This is the most favorable result, associated with the highest chance of long-term survival without recurrence.
- RCB-I (minimal residual disease) — Very little cancer remains.
- RCB-II (moderate residual disease) — A moderate amount of cancer remains.
- RCB-III (extensive residual disease) — A large amount of cancer remains. Higher risk of recurrence; additional (adjuvant) treatment after surgery is often recommended.
For HER2-positive and triple-negative breast cancers in particular, the degree of pathologic response to neoadjuvant therapy is one of the strongest predictors of long-term outcome.
Pathologic stage (pTNM)
The pathologic stage describes how far the cancer has spread based on examination of the surgical specimen. It uses the TNM staging system, which considers the primary tumor (T), lymph node involvement (N), and distant metastasis (M). The pathologist determines the pT and pN stages; the M stage is determined by imaging.
Tumor stage (pT)
- pT0 — No evidence of primary tumor found in the surgical specimen (may occur after complete response to neoadjuvant therapy).
- pT1mi — Tumor 1 mm or smaller.
- pT1a — Tumor larger than 1 mm but 5 mm or smaller.
- pT1b — Tumor larger than 5 mm but 10 mm or smaller.
- pT1c — Tumor larger than 10 mm but 20 mm or smaller.
- pT2 — Tumor larger than 20 mm but 50 mm or smaller.
- pT3 — Tumor larger than 50 mm.
- pT4a — Tumor has grown into the chest wall (excluding the pectoralis muscles alone).
- pT4b — Tumor has spread to the skin, causing ulceration or satellite skin nodules.
- pT4c — Both pT4a and pT4b.
- pT4d — Inflammatory breast cancer (redness and swelling of the entire breast skin).
Nodal stage (pN)
- pN0 — No cancer in any lymph nodes examined.
- pN0(i+) — Isolated tumor cells only (clusters ≤0.2 mm) — not counted as positive.
- pN1mi — Micrometastases only (0.2–2 mm) in axillary lymph nodes.
- pN1a — Cancer in 1–3 axillary lymph nodes, with at least one deposit larger than 2 mm.
- pN1b — Cancer in ipsilateral internal mammary sentinel nodes (excluding isolated tumor cells).
- pN2a — Cancer in 4–9 axillary lymph nodes.
- pN2b — Cancer in internal mammary lymph nodes without axillary involvement.
- pN3a — Cancer in 10 or more axillary lymph nodes, or in infraclavicular (level III axillary) lymph nodes.
- pN3b — Cancer in internal mammary lymph nodes and axillary lymph nodes.
- pN3c — Cancer in supraclavicular (above the collarbone) lymph nodes.
What is the prognosis for invasive ductal carcinoma?
The prognosis for invasive ductal carcinoma depends on several factors working together. No single finding tells the complete story — your doctor will consider all of the following:
- Stage — The most important predictor. Cancers found early and confined to the breast (pT1, pN0) have a 5-year survival rate exceeding 95%. When cancer has spread to lymph nodes or distant organs, outcomes vary more widely.
- Grade — Higher-grade tumors (grade 3) grow faster and carry a higher risk of recurrence than grade 1 tumors.
- Hormone receptor and HER2 status — ER/PR-positive tumors generally respond well to hormone therapy and have a favorable outlook. HER2-positive cancers are more aggressive but respond well to HER2-targeted treatments. Triple-negative cancers are harder to treat and have a higher short-term recurrence risk. However, they can still be curable, especially when a complete pathologic response is achieved with neoadjuvant chemotherapy.
- Lymphovascular invasion — Its presence increases the risk of lymph node involvement and distant spread.
- Surgical margins — Negative margins with adequate distance reduce the risk of local recurrence.
- Genomic test results — Tests such as the 21-gene recurrence score provide additional information about the risk of recurrence beyond standard pathologic features, particularly for hormone receptor-positive, HER2-negative cancers.
- Response to neoadjuvant therapy — Achieving a pathologic complete response (RCB-0) is associated with significantly better long-term outcomes, particularly for HER2-positive and triple-negative breast cancers.
Questions to ask your doctor
Your pathology report contains important information that will guide your care. The following questions may help you prepare for your next appointment.
- What was the size and Nottingham grade of my tumor?
- What is the pathologic stage of my cancer (pT and pN)?
- Were any lymph nodes involved, and if so, how many and what size were the deposits?
- Were the surgical margins clear? Was the tumor completely removed?
- Is lymphovascular invasion present?
- What are my hormone receptor (ER and PR) results, and do they affect my treatment?
- What is my HER2 status — negative, low, ultralow, or positive — and does it change my treatment options?
- Is my tumor triple-negative, and what does that mean for treatment?
- Was DCIS found alongside the invasive cancer, and does its extent affect surgery recommendations?
- Will I need genomic testing (such as Oncotype DX or MammaPrint), and how would the result change my treatment?
- If I received neoadjuvant therapy, what was my residual cancer burden score, and does it affect my next steps?
- Should I be referred for genetic counseling to evaluate for BRCA1/2 or other inherited mutations?
- What additional treatment is recommended — surgery, radiation, chemotherapy, hormone therapy, targeted therapy, or a combination?
- What follow-up schedule and surveillance imaging will I need?



